Moscovium is in recognition of the Moscow region and honors the ancient Russian land that is the home of the Joint Institute for Nuclear Research, where the discovery experiments were conducted using the Dubna Gas-Filled Recoil Separator in combination with the heavy ion accelerator capabilities of the Flerov Laboratory of Nuclear Reactions. These are in line with tradition, honoring a place or geographical region, and are proposed jointly by the discoverers at the Joint Institute for Nuclear Research, Dubna (Russia), Oak Ridge National Laboratory (USA), Vanderbilt University (USA), and Lawrence Livermore National Laboratory (USA). Keeping with tradition, newly discovered elements can be named after:įor the element with atomic number 115, the name proposed is moscovium with the symbol Mc and for element with atomic number 117, the name proposed is tennessine with the symbol Ts. The guidelines for naming the elements were recently revised and shared with the discoverers to assist in their proposals. A five-month public review is set, expiring 8 November 2016, prior to the formal approval by the IUPAC Council. The IUPAC Inorganic Chemistry Division has reviewed and considered these proposals and recommends these for acceptance. Oganesson and symbol Og, for the element with Z = 118. Tennessine and symbol Ts, for the element with Z = 117, Moscovium and symbol Mc,for the element with Z = 115, 
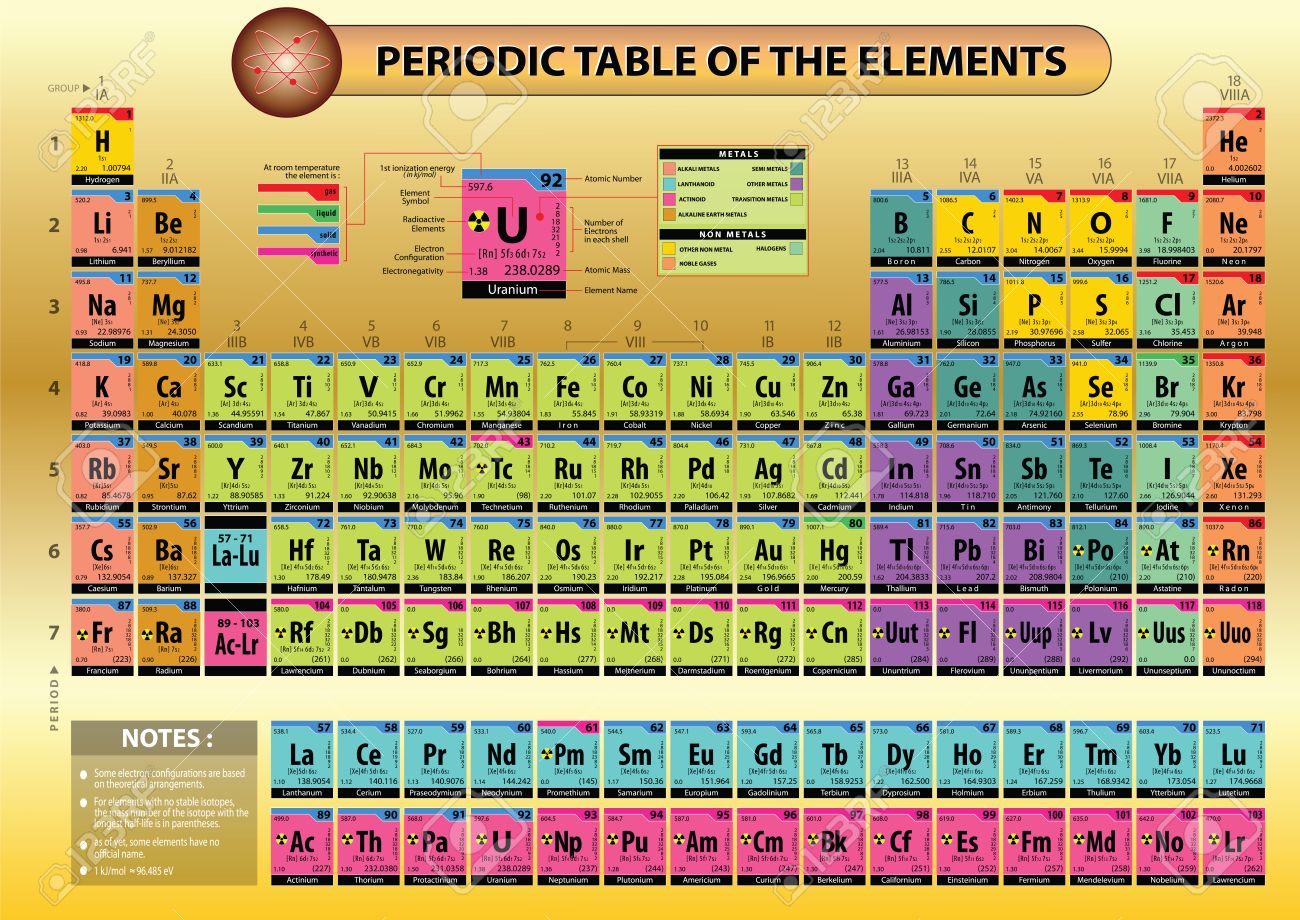
Nihonium and symbol Nh, for the element with Z = 113, Full text is available online.įollowing earlier reports that the claims for discovery of these elements have been fulfilled, the discoverers have been invited to propose names and the following are disclosed for public review: As with Chemistry, the text of Wikipedia is available under the GNU Free Documentation License.Provisional Recommendations are drafts of IUPAC recommendations on terminology, nomenclature, and symbols, made widely available to allow interested parties to comment before the recommendations are finally revised and published in IUPAC’s journal Pure and Applied Chemistry. The list of authors can be seen in the page history. The original article was at List of elements by atomic mass. Series: Alkalis - Alkaline earths - Lanthanides - Actinides - Transition metals - Poor metals - Metalloids - Nonmetals - Halogens - Noble gasesīlocks: s-block - p-block - d-block - f-block - g-block Name | Atomic symbol | Atomic number | Boiling point | Melting point | Density | Atomic mass Standard table | Vertical table | Table with names | Names and atomic masses (large) | Names and atomic masses (small) | Names and atomic masses (text only) | Inline F-block | Elements to 218 | Electron configurations | Metals and nonmetals | Table by blocks | Alternatives Atomic weights of elements with atomic numbers 110-116 taken from this source. IUPAC Standard Atomic Weights Revised (2005).Atomic weights of elements with atomic numbers from 1-109 taken from this source. Atomic Weights of the Elements 2001, Pure Appl.Note 5: The atomic weight of commercial Lithium can vary between 6.939 and 6.996-analysis of the specific material is necessary to find a more accurate value.Note 4: The isotopic composition varies in terrestrial material such that a more precise atomic weight can not be given.Note 3: The isotopic composition of the element can vary in commercial materials, which can cause the atomic weight to deviate significantly from the given value. /PeriodicTableSigFig-NoBG-56a12da75f9b58b7d0bcd00f.png)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
